| Method no.: | 36 |
| Matrix: | Air |
| Target concentration: | 10 ppm (18 mg/m3) (OSHA PEL) |
| Procedure: | Samples are collected by drawing known volumes of air through standard size sampling tubes containing XAD-7 resin coated with 10% NBD chloride by weight. The samples are desorbed with 5% (w/v) NBD chloride in tetrahydrofuran (with a small amount of sodium bicarbonate present), heated in a hot water bath, and analyzed by high-performance liquid chromatography using a fluorescence or visible detector. |
| Recommended air volume and sampling rate: |
10 L at 0.2 L/min |
| Reliable quantitation limit: | 16 ppb (29 µg/m3) |
| Standard error of estimate at the target concentration: (Figure 4.4.1.) |
8.0% |
| Status of method: | Evaluated method. This method has been subjected to the established evaluation procedures of the Organic Methods Evaluation Branch. |
| Date: May 1982 | Chemist: Carl J. Elskamp |
OSHA Analytical Laboratory
Salt Lake City, Utah
1. General Discussion
- 1.1. Background
- 1.1.1. History
The recommended air sampling procedure for ethylamine in the OSHA Field Operations Manual is collection in midget impingers containing sulfuric acid (Ref. 5.1.). The recommended air sampling procedure in NIOSH Methods 221 and S144 (Refs. 5.2. and 5.3.) requires silica gel sampling tubes. In all of these methods, the recommended analysis is done by gas chromatography. The shortcomings of these methods include: impingers are inconvenient to use in the field, stability problems appear to exist for aliphatic amines adsorbed on silica gel, and the nature of volatile aliphatic amines results in several disadvantages when analyzed by gas chromatography (peak tailing, ghosting, and column decomposition from injection of aqueous solutions). (Ref. 5.4.) These problems do not exist for this procedure where a stable NBD chloride derivative is formed on NBD chloride coated XAD-7.
The derivative is extracted with 5% (w/v) NBD chloride in tetrahydrofuran (THF) and analyzed by high-performance liquid chromatography. A similar procedure has been successfully evaluated for the collection and analysis of dimethylamine (OSHA Method 34) (Ref. 5.4.) and may be applicable for collection and analysis of other volatile aliphatic amines.
This procedure differs from OSHA Method 34 in that the analytical procedure had to be modified. It was found that a higher yield of derivative could be obtained if samples were desorbed and standards were prepared in a 5% solution of NBD chloride in tetrahydrofuran (THF) instead of a 0.4% solution as specified in the dimethylamine method. Also, it was found that the samples and standards must be heated at 60°C in a hot water bath for 2.5 h to insure a complete reaction. These two modifications did not affect the yield for the dimethylamine derivative. Thus, samples for these two analytes could be analyzed simultaneously with this procedure.
1.1.2. Toxic effects
(This section is quoted directly from "Occupational Health Guidelines for Chemical Hazards" (Ref. 5.5.) and is for information only and should not be taken as the basis of OSHA policy).
- "Ethylamine vapor is a primary irritant to mucous membranes,
eyes, and skin. Exposure to 8000 ppm for 4 h was lethal to rats.
Rabbits survived exposures to 50 ppm daily for 6 weeks but showed
pulmonary irritation and some myocardial degeneration; corneal
damage was observed after 2 weeks of exposure. In the rabbit eye,
1 drop of a 70% solution of ethylamine caused immediate, severe
irritation. Eye irritation and corneal edema in humans have been
reported from industrial exposure. A 70% solution of the base
dropped on the skin of guinea pigs caused prompt skin burns
leading to necrosis; when held in contact with guinea pig skin for
24 h there was severe skin irritation with extensive necrosis and
deep scarring."
1.1.3. Potential workplace exposure
Following are some common operations in which exposure to ethylamine may occur as reported in "Occupational Health Guidelines for Chemical Hazards." (Ref. 5.5.)
Ethylamine can be used:
- in synthesis of agricultural chemicals for herbicides; as a
dyestuff intermediate; as a solvent for dyes, resins, and oils; as
a catalyst for polyurethane foams; in pharmaceuticals, emulsifying
agents; as a vulcanization accelerator for
as a stabilizer for rubber latex; as a catalyst for curing epoxy resins.
in synthesis of dimethylolethyltriazone, a wash and wear agent for cotton fabrics; in synthesis of 1,3-diethylthiourea, a corrosion inhibitor.
in synthesis of chemical intermediates and solvents; as plasticizers and in refining of lubricating oils.
as a selective solvent in refining petroleum and vegetable oils.
in synthesis of alkyl isocyanates for intermediates in manufacture of organic products such as pharmaceuticals and resins.
in synthesis of rhodamine dyes.
as a deflocculating agent in ceramics industry; in manufacture of detergents.
1.1.4. Physical properties (Ref. 5.5.)
| molecular weight: | 45.1 |
| boiling point (760 mm Hg): | 16.7°C |
| color: | colorless liquid or gas |
| specific gravity (water = 1): | 0.7 |
| vapor pressure: | 1.18 atmospheres at 20°C |
| flash point: | <-18°C (closed cup) |
| odor: | ammonia-like |
| flammable limits in air, % by volume: |
lower: 3.5; upper: 14.0 |
| autoignition temperature: | 385°C |
| molecular formula: | C2H5NH2 |
1.2. Limit defining parameters (The air concentrations listed throughout this method are based on an air sample volume of 10 L and a desorption volume of 2.0 mL.)
- 1.2.1. Detection limit of the analytical procedure
The detection limit of the analytical procedure is 1.8 ng per injection. This is the amount of ethylamine which will give a peak whose height is approximately 5 times baseline noise. (Section 4.1.)
1.2.2. Detection limit of the overall procedure
The detection limit of the overall procedure is 0.29 µg per sample (16 ppb or 29 µg/m3). This is the calculated amount of ethylamine spiked on the sampler which allows recovery of an amount of ethylamine equivalent to the detection limit of the analytical procedure. (Section 4.2.)
1.2.3. Reliable quantitation limit
The reliable quantitation limit is 0.29 µg per sample (16 ppb or 29 µg/m3). This is the smallest amount of analyte which can be quantitated within the requirements of at least 75% recovery and a precision (±1.96 SD ) of ±25% or better. (Section 4.2.)
The reliable quantitation limit and detection limits reported in the method are based upon optimization of the instrument for the smallest possible amount of analyte. When the target concentration of an analyte is exceptionally higher than these limits, they may not be attainable at the routine operating parameters.
- 1.2.4. Sensitivity
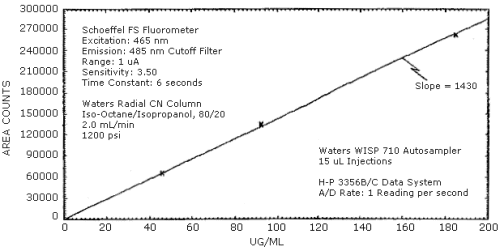
The sensitivity of the analytical procedure over the concentration range of 5 to 20 ppm is 1430 area counts per µg ethylamine/mL. The sensitivity is determined by the slope of the calibration curve. (Section 4.3.) The sensitivity will vary somewhat with the particular instrument used in the analysis.
1.2.5. Recovery
The recovery of analyte from the collection medium during storage must be 75% or greater. The recovery of ethylamine from samples used in a 15-day storage test remained above 87% when samples were stored at ambient or refrigerated temperatures. (Section 4.4.)
1.2.6. Precision (analytical method only)
The pooled coefficient of variation obtained from replicate determinations of analytical standards equivalent to 5, 10, and 20 ppm is 0.004. (Section 4.3.)
1.2.7. Precision (overall procedure)
The overall procedure must provide results at the target
concentration that are ±25% or better at the 95% confidence level.
The precision at the 95% confidence level for a
1.2.8. Reproducibility
Six samples generated on a vapor generator and a draft copy of this procedure were given to a chemist unassociated with this work. The six samples (which had been stored 21 days) were analyzed and the results reported. The average recovery was 97.9% with a standard deviation of 2.3%. (Section 4.5.)
1.3. Advantages
- 1.3.1. The solid sorbent tube provides a convenient method for
sampling.
1.3.2. Ethylamine is analyzed as a derivative which is specific, stable, and easier to quantitate than the free amine.
1.3.3. The analysis is rapid, sensitive, and precise.
1.3.4. This method is applicable to dimethylamine and possibly other volatile aliphatic amines.
1.4. Disadvantages
- 1.4.1. The method has not been field tested.
1.4.2. Sampling tubes (XAD-7 coated with 10% NBD chloride) are not commercially available.
2. Sampling Procedure
- 2.1. Apparatus
- 2.1.1. Samples are collected by use of a personal sampling pump
that can be calibrated to within ±5% of the recommended flow rate
with the sampling tube in line.
2.1.2. Samples are collected on solid sorbent sampling tubes containing XAD-7 coated with 10% NBD chloride by weight. The tube consists of two sections of coated XAD-7 separated by a glass wool plug. The front section contains 80 mg of coated sorbent and the back section, 40 mg. The sections are held in place with glass wool plugs in a glass tube 4-mm i.d. × 70-mm length.
The coated XAD-7 is prepared by rinsing the 20/50 mesh resin several times with methyl alcohol to remove fines. The resin is extracted for 24 h with methyl alcohol in a Soxhlet Extractor and dried by vacuum. The dried resin is then coated with 10% NBD chloride by weight using methylene chloride as a solvent. The solvent is removed by rotary evaporation.
2.2. Reagents
None required
2.3. Technique
- 2.3.1. Connect the sampling tube to the sampling pump with
flexible tubing. Air being sampled should not pass through any hose
or tubing before entering the sampling tube.
2.3.2. Place the sampling tube vertically in the employees' breathing zone.
2.3.3. Seal the tubes immediately after sampling with plastic caps.
2.3.4. Submit at least one blank for each sample set. The blank should be handled in the same manner as samples, except no air is drawn through it.
2.3.5. Record sample volume (in liters of air) for each sample, along with any potential interferences.
2.3.6. Ship any bulk sample(s) in a separate container(s) from the air samples.
2.4. Breakthrough
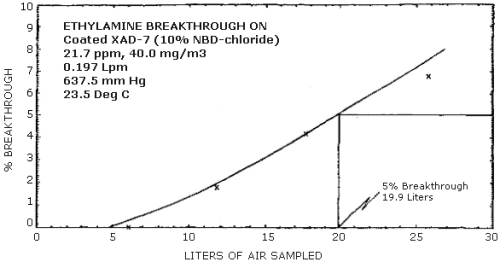
The average 5% breakthrough volume from a test atmosphere (air at about 80% relative humidity) containing 21.7 ppm (40.0 mg/m3) ethylamine was determined to be 22.4 L. This corresponds to loading of 0.896 mg on the sampling tube. The sampling rate was approximately 0.2 L/min and the test atmosphere was at 23.5°C and 637.5 mm Hg. (Section 4.6.)
2.5. Desorption efficiency
The desorption efficiency of ethylamine from spiked sample tubes was 82.7% over the range of 5 to 20 ppm. (Section 4.7.)
2.6. Recommended air volume and sampling rate
- 2.6.1. The recommended air volume is 10 L.
2.6.2. The recommended sampling rate is 0.2 L/min.
2.7. Interferences
- 2.7.1. An interference study was performed in which 10-L air
samples of a test atmosphere containing approximately 10 ppm each of
ethylamine, methylamine, and dimethylamine were collected. The test
atmosphere was at about 80% relative humidity. There was no
difference in the amount of ethylamine derivative recovered whether
the other amines were present or not.
2.7.2. It is not known if any compound(s) will interfere with the collection of ethylamine on coated XAD-7 tubes.
2.7.3. Suspected interferences should be reported to the laboratory with submitted samples.
2.8. Safety precautions
- 2.8.1. Attach the sampling equipment to the employee so that it
will not interfere with work performance or safety.
2.8.2. Follow all safety procedures that apply to the work area being sampled.
3. Analytical Procedure
- 3.1. Apparatus (The particular apparatus used for this study can
be found in Figure 4.3.)
- 3.1.1. High-performance liquid chromatograph (HPLC) equipped
with a fluorescence and/or visible detector.
3.1.2. An HPLC column capable of separating the ethylamine derivative from NBD chloride and any interferences. A Radial CN column was used in this study in the normal phase since the NBD chloride derivatives fluoresce stronger in nonaqueous solvent systems. Reduced sensitivities with fluorescence detection will be obtained with the use of an aqueous reverse phase column system.
3.1.3. An electronic integrator or some other suitable method of measuring peak areas or heights.
3.1.4. A mechanical shaker.
3.1.5. A hot water bath.
3.1.6. Volumetric flasks for preparing standards and making dilutions.
3.1.7. Pipets and syringes for preparing standards, making dilutions, and dispensing reagents.
3.1.8. Small vials with Teflon-lined caps, capable of holding 3 mL.
3.2. Reagents
- 3.2.1. HPLC grade isopropanol and isooctane.
3.2.2. Reagent grade tetrahydrofuran. (THF)
3.2.3. Reagent grade sodium bicarbonate.
3.2.4. Ethylamine solution in water of known concentration or ethylamine gas.
3.2.5. Reagent grade NBD chloride (7-chloro-4-nitrobenzo-2-oxa1,3-diazole).
3.2.6. Desorption reagent: 5 g of NBD chloride per 100 mL of THF.
3.3. Standard preparation
- 3.3.1. Prepare a stock standard of ethylamine by diluting a
known volume of ethylamine with THF.
3.3.2. Working standards are prepared by injecting microliter amounts of the stock standard into 2.0 mL of desorption reagent in a small vial. Example: If a 5-µL aliquot of a stock standard at a concentration of 37 µg/µL is injected into a vial containing 2.0 mL of NBD chloride solution, the working standard is equal to 185 µg/sample (5 × 37) if the samples are desorbed with 2.0 mL. For a 10-L air sample this is equivalent to 18.5 mg/m3, uncorrected for desorption efficiency.
3.3.3. Add approximately 25 mg of solid sodium bicarbonate to each vial and seal with Teflon-lined caps. (This is easily done by using the large end of a standard size disposable dropping pipette as a spatula). The standards are shaken for 0.5 h and then heated for 2.5 h at 60°C in a water bath. Allow standards to cool to room temperature before analyzing.
3.4. Sample preparation
- 3.4.1. Transfer each section of the sample to separate vials.
The glass wool plugs must be added to the vials if they contain
entrapped XAD-7 beads. The glass tube is discarded.
3.4.2. Add 2.0 mL of the desorption reagent.
3.4.3. Add approximately 25 mg of solid sodium bicarbonate to each vial.
3.4.4. Seal the vials with Teflon-lined caps and shake in a horizontal position for 0.5 h. The vials should be positioned parallel to the shaker's movement.
3.4.5. Heat the vials for 2.5 h in a hot water bath at 60°C. Cool to room temperature before analyzing.
3.5. Analysis
- 3.5.1. HPLC conditions
| fluorescence detector: | 465 nm excitation 525 nm emission |
| injection size: | 15 µL |
| column: | Waters Radial CN |
| solvent: | isooctane:isopropanol, 80:20 at 2 mL/min |
| retention time of ethylamine derivative: |
3.3 min |
| alternate detector: | Visible at 465 nm |
| chromatograms: | Section 4.8. |
3.5.2. Peak areas (or heights) are measured by an integrator or other suitable means.
3.5.3. A calibration curve is constructed by plotting peak areas (or heights) of standard injections versus µg per sample. Sample concentrations must be bracketed by standards.
3.6. Interferences
- 3.6.1. Any compound that has the same general retention time as
the ethylamine derivative and responds with the detector used is an
interference. Possible interferences should be reported to the
laboratory with submitted samples by the industrial hygienist. The
derivatives of methylamine, dimethylamine, and diethylamine can be
separated from the ethylamine derivative.
3.6.2. HPLC parameters (i.e. solvent composition, column, detector, etc.) may be changed to circumvent interferences.
3.6.3. Retention time on a single column is not considered proof of chemical identity. Samples over the PEL should be confirmed by GC/MS or other suitable means.
3.7. Calculations
The ethylamine concentration is obtained from the calibration curve in terms of micrograms per sample. The air concentration for samples is calculated using the following formulae. If any ethylamine derivative is found on the backup section, it is added to the amount found on the front section. The total amount is then corrected by subtracting the total amount found in the blank.
| mg/m3 = | (blank-corrected micrograms per
sample)
(liters of air sampled) (desorption efficiency) |
ppm = (mg/m3)(24.46)/(45.1) = (mg/m3)(0.542)
| where | 24.46 | = | molar volume (liters) at 25°C and 760 mm Hg |
| 45.1 | = | molecular weight of ethylamine |
3.8. Safety precautions
- 3.8.1. Avoid skin contact and inhalation of all chemicals used,
especially ethylamine and NBD chloride.
3.8.2. Restrict the use of all chemicals to a fume hood if possible.
3.8.3. Wear safety glasses and lab coat at all times while in the laboratory area.
4. Backup Data
- 4.1. Detection limit of the analytical procedure
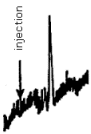
The detection limit of the analytical procedure is 1.8 ng/injection. This was determined by making a 15-µL injection of a 0.123 µg/mL standard. This standard is based on the weight of ethylamine added to make the standard. Shown in Figure 4.1. is a chromatogram obtained from a Schoeffel FS 970 Fluorescence detector set at 0.02-µA range, 4.75 sensitivity, and 6-s time constant. The recorder was set at 0.2 cm/min and 10-mV full scale.
4.2. Detection limit of the overall procedure and reliable quantitation limits.
Samples were prepared by spiking (liquid injection) the coated XAD-7 with an amount of ethylamine equivalent to the analytical detection limit (0.246 µg/sample). The samples were allowed to sit overnight before being extracted and analyzed.
Detection Limit of the Overall Procedure
and Reliable Quantitation Limit Data
|
| |||
| % recovery | statistics | ||
|
| |||
| 87.0 82.6 87.0 |
SD |
= = |
85.5 2.9 |
|
| |||
The detection limit of the overall procedure and the reliable quantitation limit is:
| 0.246 µg/sample
0.855 |
= 0.29 µg/sample |
4.3. Sensitivity and precision
The sensitivity and precision of the analytical procedure were obtained from multiple injections of analytical standards. This data is given in Table 4.3. and shown graphically in Figure 4.3.
Sensitivity and Precision Data
|
| |||
| × target conc. µg/mL |
0.5× 46.2 |
1× 92.4 |
2× 184.8 |
|
| |||
| area
counts SD CV |
67002 66134 66408 66540 66339 66014 66406.2 347.9 0.0052 |
135865 135010 134995 135139 135684 136740 135572 678.1 0.0050 |
262363 261833 262413 262819 263364 262969 262627 537.2 0.0020 |
|
| |||
4.4. Recovery and storage data
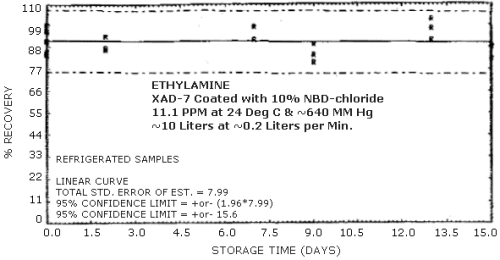
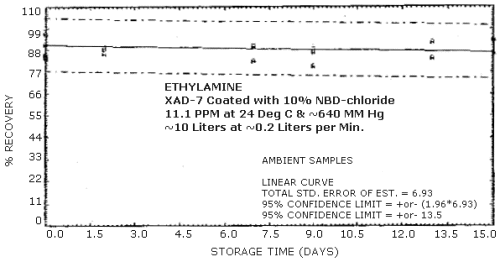
Storage samples were generated from a test atmosphere (air) containing 11.1 ppm ethylamine at approximately 80% relative humidity, 24°C, and 640 mm Hg. Each sample was generated by sampling the test atmosphere at approximately 0.2 L/min for 50 min, resulting in a sample volume of about 10 L. An amount of coated XAD-7 equivalent to the front section of a standard adsorbent tube (about 80 mg) was used for each sample. After sampling, the adsorbent was transferred to separate WISP vials, capped, and stored. Six samples were extracted and analyzed immediately after generation, 15 were stored in a closed drawer at ambient temperature, and 15 were stored under refrigeration at 0°C. The results of these data are shown graphically in Figures 4.4.1. and 4.4.2.
Storage Tests
|
| |||||||
| storage time | % recovery | ||||||
| (days) | (refrigerated) | (ambient) | |||||
|
| |||||||
| 0 0 2 7 9 13 15 |
99.5 84.2 93.6 98.2 80.6 102.2 88.9 |
85.4 96.0 87.8 ---1 84.3 97.6 82.4 |
96.4 91.1 86.5 91.9 89.7 91.8 88.2 |
99.5 84.2 86.4 82.6 87.1 84.1 86.6 |
85.4 96.0 89.0 90.1 80.0 ---1 93.3 |
96.4 91.1 89.5 90.0 90.0 92.1 86.7 | |
|
| |||||||
| 1 Samples lost; orifice plugged during sampling. | |||||||
4.5. Reproducibility
Six ethylamine samples were generated on the vapor generator by sampling from an air stream containing 11.1 ppm ethylamine for 50 min at approximately 0.2 L/min. The sample stream was at 24.8°C, 640 mm Hg, and approximately 80% relative humidity. The samples were stored for 21 days at room temperature before being analyzed.
Reproducibility
|
| ||||||
| sample no. | mg found | mg expected | % found | |||
|
| ||||||
| 1 2 3 4 5 6 7 (blank) |
0.2047 0.1060 0.2020 0.2123 0.2097 0.2202 N.D. |
0.2115 0.2368 0.2097 0.2150 0.2065 0.2292 0.00 |
96.8 (44.8)1 96.3 98.7 101.5 96.1 --- | |||
| ||||||
|
| ||||||
| 1 This sample was not used for calculation of average % found. The orifice plugged during sampling, resulting in a lower sampling rate. | ||||||
4.6. Breakthrough
The breakthrough volume was determined from a test atmosphere containing 21.7 ppm (40.0 mg/m3) ethylamine. The sampling tube contained only the front section (about 80 mg) of adsorbent. A backup tube was connected downstream from the sampling tube. This backup tube was changed periodically and analyzed to determine the amount of ethylamine breaking through the sampling tube. The average breakthrough volume for three separate determinations was 22.4 L. This corresponds to an average loading of 0.896 mg of ethylamine on the sample tube when 5% breakthrough occurs. The test atmosphere was at 23.5°C, 637.5 mm Hg, and about 80% relative humidity. Given in Table 4.6. and shown graphically in Figure 4.6. is the results from one of the breakthrough determinations. The 5% breakthrough volume was 19.9 L for this determination.
Breakthrough Data
|
| |||
| minutes1 | liters | mg founds2 | % breakthrough |
|
| |||
| 30 60 90 131 |
5.9 11.81 17.71 25.80 |
0.0 8.5 29.5 70.6 |
0.0 1.8 4.2 6.8 |
|
| |||
| 1
time backup tube was changed 2 cumulative amounts | |||
4.7. Desorption efficiency
Desorption efficiencies were determined by injecting known amounts of analyte onto coated XAD-7, with analysis the next day.
Desorption Efficiency
|
| |||
| × target conc. µg/sample |
0.5× 92.4 |
1× 184.8 |
2× 369.6 |
|
| |||
| desorption efficiency, % |
86.5 84.6 85.6 83.4 83.4 85.4 84.8 |
82.7 82.8 79.8 82.2 81.6 83.7 82.1 |
80.4 80.4 lost lost 80.1 80.7 80.4 |
|
| |||
4.8. Chromatograms
Chromatograms of an ethylamine standard are shown in Figure 4.8. The chromatograms are from a 15-µL injection of a 0.185 mg ethylamine per sample standard. The fluorescence and visible detectors were connected in series to give essentially simultaneous chromatograms. The response shown is standardized to keep the peaks about 90% full scale. This figure should not be interpreted to determine the relative response of each detector.
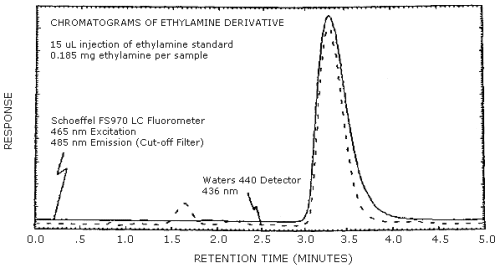
5. References
- 5.1. "Industrial Hygiene Field Operation Manual", OSHA Instruction
CPL 2-2.20, Office of Field Coordination, 1979.
5.2. "NIOSH Manual of Analytical Methods", Vol. 1, 2nd Edition, April 1977, USDHEW, PHS, CDC, NIOSH, DHEW (NIOSH) Publication No. 77-157-A, Method P&CAM 221.
5.3. "NIOSH Manual of Analytical Methods", Vol. 3, 2nd Edition, April 1977, USDHEW, PHS, CDC, NIOSH, DHEW (NIOSH) Publication No. 77-157-C, Method S144.
5.4. Elskamp, C.J., Dimethylamine (Method 34, Organic Methods Evaluation Branch, OSHA Analytical Laboratory, Salt Lake City, Utah). Unpublished (2-82).
5.5. "Occupational Health Guidelines for Chemical Hazards", NIOSH/OSHA, Jan. 1981, DHHS (NIOSH) Publication No. 81-123.