ACRYLAMIDE
| Method number: | PV2004 |
| Matrix: | Air |
| Target Concentration: | 0.03 mg/m3 (OSHA PEL) |
| Procedure: | Samples are collected by drawing known volumes of air through OSHA versatile sampler (OVS-7) tubes, each containing a glass fiber filter and two sections of XAD-7 adsorbent. Samples are desorbed with a solution of 5% methanol 95% water and analyzed by high performance liquid chromatography (HPLC) using an ultraviolet detector (UV). |
| Recommended air volume and sampling rate: |
120 L at 1.0 L/min |
| Detection limit of the overall procedure (based on the recommended air volume and the analytical detection limit): |
0.00125 mg/m3 |
| Status of method: | Stopgap method. This method has been partially evaluated and is presented for information and trial use only. |
| Date: July 1991 (final) | Chemist: Duane Lee |
Organic Service Branch II
OSHA Salt Lake Technical
Center
Salt Lake City, Utah
1. General Discussion
- 1.1. Background
- 1.1.1. History of procedure
The necessity for additional information on the sampling and analysis of acrylamide became apparent because of several factors. Since the development of the acrylamide method, different kinds of samplers have become available. The sensitivity of the method needed to be checked, because the PEL has been changed from 0.3 mg/m3 to 0.03 mg/m3. Also, there were some questions about the stability of acrylamide on a sampler when exposed to elevated temperatures.
The sensitivity of the existing method was investigated because of the lower PEL. (Ref. 5.1.) After reviewing the method it appeared to have a detection limit around one seventh of the PEL based on the recommended air volume and desorption volume. Further investigation was done on a gas chromatograph and the detection limit was found to be 0.7 µg/mL, which calculates to be 0.006 mg/m3 for a 1-mL desorption volume or 0.029 mg/m3 for a 5-mL desorption volume based on a 120 L air volume. This level was insufficient for use with OVS type samplers and for air volumes less than 120 L. Therefore a published liquid chromatography procedure was tried which yielded better sensitivity. (Ref. 5.3.)
The stability of acrylamide was tested on the OVS samplers with
different types of resins. Several front sections of various types
of the OVS sampler configuration were tested for the stability of
acrylamide. The types tested were
After storage the samples were desorbed with 2 mL of methanol and analyzed by the gas chromatography method. The results are listed in the following tables.
OVS-2 Room Temperature
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| #1 #2 #3 #4 #5 #6 |
4.38 4.38 4.38 4.38 4.38 4.38 |
4.81 4.80 4.06 4.73 4.24 3.90 |
109.8 109.6 92.6 108.1 96.8 89.1 |
| Average = 101.0 | |||
OVS-2 Oven Temperature (45°C)
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| #1 #2 #3 #4 #5 #6 |
4.38 4.38 4.38 4.38 4.38 4.38 |
3.95 4.12 4.63 4.33 4.28 4.14 |
90.2 95.0 105.8 98.9 97.7 94.6 |
| Average = 97.0 | |||
OVS-7 Room Temperature
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| #1 #2 #3 #4 #5 #6 |
4.38 4.38 4.38 4.38 4.38 4.38 |
3.94 4.60 3.81 4.71 3.28 4.70 |
89.9 104.9 87.0 107.5 74.9 107.3 |
| Average = 95.2 | |||
OVS-7 Oven Temperature (45°C)
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| #1 #2 #3 #4 #5 #6 |
4.38 4.38 4.38 4.38 4.38 4.38 |
4.23 3.77 4.20 4.67 4.36 4.03 |
96.6 86.0 95.9 106.6 99.5 92.0 |
| Average = 96.1 | |||
OVS-SG Room Temperature
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| #1 #2 #3 #4 #5 #6 |
4.38 4.38 4.38 4.38 4.38 4.38 |
5.46 4.33 4.44 4.61 4.13 4.39 |
124.6 98.9 101.5 105.3 94.4 100.2 |
| Average = 104.2 | |||
OVS-SG Oven Temperature (45°C)
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| #1 #2 #3 #4 #5 #6 |
4.38 4.38 4.38 4.38 4.38 4.38 |
4.78 4.57 4.03 3.92 3.25 3.99 |
109.2 104.3 92.1 89.6 74.2 91.20 |
| Average = 93.4 | |||
The above data shows that acrylamide is stable on the different resins for four days at 45°C. The stability could change at higher temperatures or by exposing the samples to UV light.
1.1.2. Toxic effects (This section is for information only and should not be taken as the basis of OSHA policy.)
Acrylamide exposure comes from inhalation and absorption through the skin. Repeated exposure to acrylamide will effect the central nervous system. Some of the symptoms from exposure are muscular weakness, ataxia, in coordination, tremors, and hallucinations. Also, there is some evidence that acrylamide is carcinogenic in rats and it is assigned to a suspect human carcinogen list. (Ref. 5.4.)
1.1.3. Potential workplace exposure
Acrylamide is a reactive monomer that is used in the production of organic chemicals. Also, it is used as a polymer or copolymer in applications as adhesives, fibers, paper sizing, molded parts, water coagulant aids, and textiles. (Ref. 5.4.) There is a potential for approximately 20,000 workers to be exposed to acrylamide. (Ref. 5.1.)
1.1.4. Physical properties (Ref. 5.1. to 5.3.)
| CAS number: | 79-06-1 |
| IMIS number: | 0115 |
| Molecular weight: | 71.08 |
| Molecular formula: | CH2CHCONH2 |
| Melting point: | 84.5°C |
| Boiling point: | 125°C (25 mm Hg) |
| Vapor pressure: | 0.9331 Pa (0.007 mm Hg) at 25°C |
| Solubility: | soluble in water, alcohol, acetone; insoluble in heptane, benzene |
| Chemical name: | acrylamide |
| Synonyms: | propenamide |
| Description: | flake like crystals; polymerizes at the melting point or under UV light |
| UV scan: | See Figure 1. |
| Structure: | 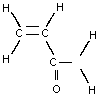 |
1.2. Limit defining parameters
The detection limit of the analytical procedure is 0.75 ng per injection. This is the amount of analyte which will give a peak whose height is approximately five times the baseline noise. (Figure 2.)
2. Sampling Procedure
- 2.1. Apparatus
- 2.1.1. A personal sampling pump that can be calibrated to within
±5% of the recommended flow rate with the sampling device in line.
2.1.2. OVS-7 tubes, which are specially made 13-mm o.d. glass tubes that are tapered to 6-mm o.d., packed with two sections of cleaned XAD-7 adsorbent and a 13-mm diameter glass fiber filter. The sampling section and backup section contain 270 and 140 mg respectively. The backup section is retained by two foam plugs and the sampling section is between a foam plug and the glass fiber filter. The glass fiber filter is held next to the sampling section by a polytetrafluoroethylene (PTFE) retainer. (Figure 2.)
2.2. Reagents
No sampling reagents are required.
2.3. Sampling technique
- 2.3.1. Immediately before sampling, remove the plastic caps from
the OVS-7 tube.
2.3.2. Attach the small end of the tube to the sampling pump with flexible tubing.
2.3.3. Attach the tube vertically in the employee's breathing zone in such a manner that it does not impede work performance.
2.3.4. After sampling for the appropriate time, remove the tube and seal it with plastic caps.
2.3.5. Wrap each sample end-to-end with an OSHA seal (Form 21).
2.3.6. Record the air volume for each sample, and list any possible interferences.
2.3.7. Submit at least one blank for each set of samples. Handle the blank in the same manner as the samples, except no air is drawn through it.
2.3.8. Submit bulk samples for analysis in a separate container. Do not ship with air samples.
2.4. Extraction efficiency
Six vials containing an amount of XAD-7 resin equivalent to the front section of an OVS-7 tube and a 13-mm glass fiber filter were each liquid-spiked with 4.003 µg of acrylamide. These samples were allowed to dry in a drawer overnight. The next day these vials were each desorbed with 5.0 mL of a solution containing 5% methanol 95% water, shaken for 30 min and then analyzed as in Section 3. The results are listed in Table 2.4.
Extraction Efficiency
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| Ex1 Ex2 Ex3 Ex4 Ex5 Ex6 |
4.003 4.003 4.003 4.003 4.003 4.003 |
3.507 3.542 3.513 3.499 3.533 3.613 |
87.6 88.5 87.89 87.4 88.3 90.3 |
| Average = 88.3 | |||
2.5. Retention efficiency
Eighteen OVS-7 tubes were liquid-spiked with 4.003 µg of acrylamide. These were allowed to equilibrate overnight in a drawer. The next day these tubes were placed on a humid air generator and 120 L of humid air (~76% relative humidity) were drawn through each tube at 1 L/min. Six of the tubes were each desorbed with 5.0 mL of a solution containing 5% methanol 95% water, shaken for 30 min and then analyzed as in Section 3. The remaining samples were stored 6 in a drawer at ambient temperature and 6 in a freezer for use in a storage study below. The results are listed in Table 2.5.
Retention Efficiency
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| R1 R2 R3 R4 R5 R6 |
4.003 4.003 4.003 4.003 4.003 4.003 |
3.651 3.665 3.661 3.665 3.782 3.934 |
91.2 91.6 91.5 94.5 98.3 |
| Average = 93.1 | |||
2.6. Sample storage
After 7 days of storage, 6 samples were each desorbed with 5.0 mL of a solution containing 5% methanol 95% water, shaken for 30 min and then analyzed as in Section 3. Three of the samples were from ambient storage and the other three were from the freezer storage samples. The remaining samples were analyzed after 13 days of storage. The results are given in Tables 2.6.1. and 2.6.2.
Ambient Storage
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| 7 7 7 13 13 13 |
4.003 4.003 4.003 4.003 4.003 4.003 |
3.635 3.664 3.625 3.487 3.536 3.417 |
90.8 91.5 90.6 87.1 88.3 85.4 |
| Average of seven days = 91.0 | |||
| Average of thirteen days = 86.9 | |||
Freezer Storage
| Amount | Amount | % | |
| Sample # | Spiked, µg | Found, µg | Recovered |
| 7 7 7 13 13 13 |
4.003 4.003 4.003 4.003 4.003 4.003 |
3.712 3.676 3.671 3.748 3.654 3.514 |
92.7 91.8 91.7 93.6 91.3 87.8 |
| Average of seven days = 92.1 | |||
| Average of thirteen days = 90.9 | |||
2.7. Recommended air volume and sampling rate
- 2.7.1. The recommended air volume is 120 L.
2.7.2. The recommended flow rate is 1.0 L/min.
2.8. Interferences (sampling)
It is not known if any compounds will interfere with the collection of acrylamide. Any suspected interferences should be reported to the laboratory.
2.9. Safety precautions (sampling)
- 2.9.1. Attach the sampling equipment in such a manner that it
will not interfere with work performance or employee safety.
2.9.2. Follow all safety practices that apply to the work area being sampled.
3. Analytical Procedure
- 3.1. Apparatus
- 3.1.1. A balance capable of weighing to the nearest tenth of a
milligram. A Mettler HL52 balance was used in this evaluation.
3.1.2. A mechanical shaker.
3.1.3. An HPLC equipped with a UV detector. A Hewlett Packard (HP) 1090M equipped with an autosampler and diode array detector was used in this evaluation.
3.1.4. An HPLC column capable of separating acrylamide from any interferences. A 25 cm × 4.6 mm i.d. Zorbax ODS (5 µm) liquid chromatography column was used in this evaluation.
3.1.5. An electronic integrator, or some other suitable means for measuring detector response. The Hewlett-Packard 1090M Data System was used in this evaluation.
3.1.6. Volumetric flasks and pipets.
3.1.7. Vials, 2-mL and 7-mL.
3.2. Reagents
- 3.2.1. Acetonitrile, HPLC grade, obtained from Burdick and
Jackson was used in this evaluation.
3.2.2. Acrylamide, reagent grade, obtained from J. T. Baker was used in this evaluation.
3.2.3. Water, HPLC grade, Milli-Q filtered water, Millipore Inc.
3.3. Standard preparation
Prepare stock standards by weighing 10 to 15 mg of acrylamide. Transfer the acrylamide to separate 10-mL volumetric flasks, and add a solution containing 5% methanol 95% water to the mark. Make working range standards of 2.0 to 185 µg/mL by diluting the stock standards with a solution containing 5% methanol 95% water. Store stock and diluted standards in a refrigerator.
3.4. Sample preparation
- 3.4.1. Transfer the glass fiber filter to a 7-mL vial.
3.4.2. Add 5.0 mL of a solution containing 5% methanol 95% water to each vial and seal with a Teflon-lined cap.
3.4.3. Shake the vials for 30 minutes on a mechanical shaker.
3.4.4. If necessary, transfer the samples to 2-mL vials for use on an HP autosampler.
3.5. Analysis
- 3.5.1. Instrument conditions
| Column: | 25 cm × 4.6 mm Zorbax ODS (5 µm) |
| Mobile phase: | 5% methanol 95% water |
| Flow rate: | 1.0 mL/min |
| Wavelength: | 200 nm, 214 nm, 240 nm |
| Retention time: | 4.0 min |
| Injection volume: | 25.0 µL |
3.5.2. Chromatogram (Figure 3.)
3.6. Interferences (analytical)
- 3.6.1. Any collected compound having a similar retention time to
that of the analyte is a potential interference.
3.6.2. HPLC conditions may generally be varied to circumvent interferences.
3.6.3. Retention time on a single column is not proof of chemical identity. Analysis on an alternate HPLC column and confirmation by mass spectrometry are additional means of identification.
3.7. Calculations
- 3.7.1. Construct a calibration curve (Figure 4.) by plotting
detector response versus concentration (µg/mL) of acrylamide.
3.7.2. Determine the µg/mL of acrylamide in each sample and blank from the calibration curve.
3.7.3. Blank correct each sample by subtracting the µg/mL found in the blank from the µg/mL found in the sample.
3.7.4. Determine the air concentration by using the following formula.
| mg/m3 = | (µg/mL, blank corrected) ×
(extraction volume, mL)
(air volume, L) × (extraction efficiency, decimal) |
3.8. Safety precautions (analytical)
- 3.8.1. Avoid skin contact and air exposure to acrylamide.
3.8.2. Avoid skin contact with all solvents.
3.8.3. Wear safety glasses at all times.
4. Recommendation for Further Study
This method should be fully validated.
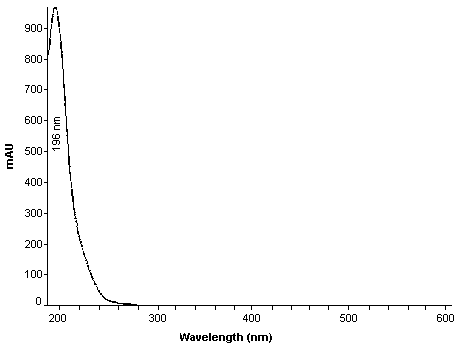
UV Scan of Acrylamide in Mobile Phase
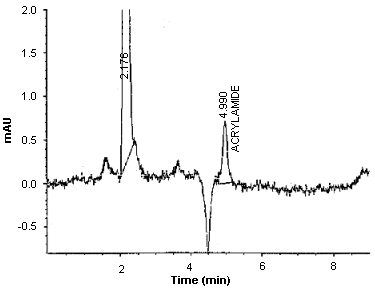
Detection Limit Chromatogram of Acrylamide at 200 nm
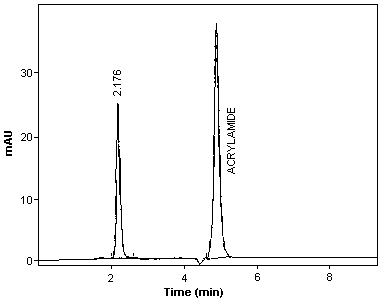
Chromatogram of Acrylamide at 200 nm
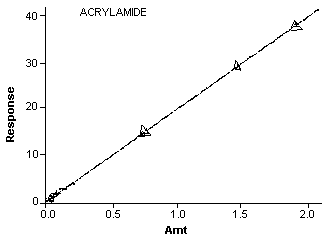
Calibration Curve
5. References
- 5.1. OSHA Analytical Methods Manual, Second Edition, U.S.
Department of Labor, Occupational Safety and Health Administration;
OSHA Analytical Laboratory: Salt Lake City, UT, 1990; Method 21;
American Conference of Governmental Industrial Hygienists (ACGIH):
Cincinnati, Oh, Publication No. 4542.
5.2. Merck Index, 10th ed.; Windholz, Martha Ed.; Merck: Rahway, NJ, 1983; p 19.
5.3. Skelly, Norman E. and Husser, Edward R.; Anal. Chem; 1978, 50, 1959-1962.
5.4. Documentation of the Threshold Limit Values and Biological Exposure Indices; American Conference of Governmental Industrial Hygienists Inc., Fifth Edition, 1986, pp. 12-13.