ACETOPHENONE
| Method number: | PV2003 |
| Matrix: | Air |
| Target concentration: | 100 ppm |
| Procedure: | Samples are collected by drawing a known volume of air through a Tenax GC tube. Samples are desorbed with a solvent mixture of 5:95 isopropanol:carbon disulfide and analyzed by gas chromatography with a flame ionization detector. |
| Recommended air volume and sampling rate: |
12.0 liters at 0.1 Lpm |
| Status of Method: | Stop gap method. This method has been only partially evaluated and is presented for information and trial use. |
| Date: 7/20/82 | Chemist: Wayne Potter |
Service Branch I
OSHA Technical Center
Salt Lake
City, Utah
1. General Discussion
- 1.1. Background
- 1.1.1. History of procedure
Recently, the OSHA Analytical Laboratory received a set of field
samples that required analysis for acetophenone. The air samples had
been collected on charcoal tubes and isopropanol impingers.
Desorption studies were done on charcoal tubes using acetone, carbon
disulfide, 5:95 isopropanol:carbon disulfide, and methylene chloride
as desorbing solvents. The best results were obtained with 5:95
isopropanol:carbon disulfide, but the recovery was only 68%, which
indicated that charcoal tubes should not be recommended for the
collection of acetophenone air samples. An evaluation of isopropanol
impingers was not performed because it was preferable to find an
adsorbent procedure for sample collection. NIOSH method 291 for
1.1.2. Toxic effects
(This section is for information only and should not be taken as the basis of OSHA policy.)
Acetophenone is a narcotic in high concentrations (Ref. 5.1). The
oral
1.1.3. Potential workplace exposure
No workplace exposure level could be found in the literature, but acetophenone is used in the following processes: perfumery, solvent, intermediate for pharmaceuticals, resin, polymerization catalyst, and flavoring (Ref.5.1)
1.1.4. Physical properties (ref. 5.1)
| Synonyms: | phenyl methyl ketone; hypnone; acetylbenzene |
| Molecular weight: | 120.15 |
| Density: | 1.0281 |
| Flash point | 82.2°C (180°F) (COC) |
| Boiling point: | 202°C |
| Freezing point: | 19.7°C |
| Solubility: | slightly soluble in water, freely soluble in alcohol, chloroform, ether, fatty oils, and glycerol. |
| Description: | Combustible, colorless liquid, with sweet, pungent odor and taste. |
| Molecular structure: | C6H5COCH3 |
1.2. Limit defining parameters
- 1.2.1. Detection limit of the analytical procedure is 20.6
ng/injection. This is the amount of analyte which will give a peak
whose height is approximately five times the baseline noise.
1.2.2. Sensitivity
The sensitivity of the analytical procedure over a concentration range of 10.28 to 1028.1 µg/mL is 407297 area units per µg/mL of acetophenone. The sensitivity is determined by the slope of the calibration curve. (See Figure 2.)
2. Sampling Procedure
- 2.1. Apparatus
- 2.1.1. A calibrated personal sampling pump whose flow can be
determined with ± 5% of the recommended flow.
2.1.2. Tenax GC tubes: glass tube with both ends flame sealed, 7 cm long with a 6 mm O.D. and a 4 mm I.D., containing 2 sections of Tenax GC separated by a 2 mm portion of urethane foam. The absorbing section contains 30 mg of Tenax GC, the backup section 15 mg. A 3 mm portion of urethane foam is placed between the outlet end of the tube and the backup section. A plug of silylated glass wool is placed in front of the absorbing section.
2.2. Reagents
- 2.2.1. None required.
2.3. Sampling technique
- 2.3.1 Immediately before sampling, break the ends of the Tenax
GC tubes. All tubes must be from the same lot.
2.3.2. Connect the tube to the sampling pump with flexible tubing. The backup section of the Tenax GC tubes should be positioned nearer the sampling pump.
2.3.3. The tubes should be placed in a vertical position during sampling to minimize channeling.
2.3.4. Air being sampled should not pass through any hose or tubing before entering the Tenax GC tubes.
2.3.5. Seal the Tenax GC tubes with plastic caps immediately after sampling. Seal each sample with official OSHA label length wise.
2.3.6. With each batch of samples, submit at least one blank tube from the same lot used for samples. This tube should be subjected to exactly the same handling as the samples (break, seal, transport) except that no air is drawn through it.
2.3.7. Transport the samples (and corresponding paperwork) to the laboratory for analysis.
2.3.8. If bulk samples are submitted for analysis, they should be
transported in glass containers with
2.4. Desorption efficiency
The average desorption efficiency from the Tenax GC tubes spiked with 1.028 mg of acetophenone was 99.1%.
| Average | |||
| Sample | Treatment | Recovery | Recovery |
| 1 | No Air | 99.51 | |
| 2 | No Air | 100.99 | 99.1 |
| 3 | No Air | 99.29 | |
| 4 | No Air | 98.25 | |
| 5 | No Air | 97.68 | |
2.5. Retention efficiency
Eight Tenax GC tubes were spiked with 1.028 mg of acetophenone. Twelve liters of dry air were drawn through four of the tubes at 0.1 Lpm. Twelve liters of humid air (about 85% relative humidity) were drawn through the other four tubes at 0.1 Lpm.
| Average | |||
| Sample | Treatment | % Recovery | % Recovery |
| 1 | 12 L Dry Air | 100.89 | |
| 2 | " | 99.79 | 99.3 |
| 3 | " | 95.72 | |
| 4 | " | 99.93 | |
|
| |||
| 5 | 12 L Humid Air | 104.50 | |
| 6 | " | 102.08 | 99.7 |
| 7 | " | l06.57 | |
| 8 | " | 85.54 | |
2.6. Sample storage
Four Tenax GC tubes were spiked with 1.028 mg of acetophenone. Two tubes were stored in a refrigerator for six days and two tubes were stored at ambient temperature for six days.
| Storage | Average | |||
| Sample | Days | Treatment | % Recovery | % Recovery |
| 1 | 6 | Refrigerator | 97.95 | |
| 2 | 6 | " | 93.55 | 95.8 |
|
| ||||
| 3 | 6 | Ambient | 96.13 | 96.7 |
| 4 | 6 | " | 97.22 | |
2.7. Recommended air volume and sampling rate.
- 2.7.1. The recommended air volume is 12.0 liters.
2.7.2. The recommended sampling rate is 0.1 liters per minute.
2.8. Interferences
- 2.8.1. It is important to be aware of other components in the
atmosphere which may interfere with the collection of the analyte.
2.9. Safety precautions
- 2.9.1. Care must be taken when opening the sealed ends of the
Tenax GC tubes to avoid serious cuts to the hands.
2.9.2. Safety glasses should be worn when opening the sealed ends of the Tenax GC tubes to avoid injury to the eyes from glass splinters.
2.9.3. Attach the sampling equipment to the worker in such a manner that it will not interfere with work performance of the employee.
2.9.4. Follow all safety practices that apply to the work area being sampled.
3. Analytical procedure
- 3.l. Apparatus
- 3.1.1. Gas chromatograph equipped with a flame ionization
detector.
3.1.2. GC column capable of separating the solvent, acetophenone and an internal standard (n-hexylbenzene). The column used for validation studies was: 10 ft × 1/8 inch stainless steel with 3% 2100 100/120 Supelcoport.
3.1.3. An electronic integrator or some other suitable method of measuring peak areas.
3.1.4. Two milliliter vials with
3.1.5. Two microliter syringes for sample injections.
3.1.6. Volumetric flasks,
3.1.7. Pipettes for dispensing desorbing reagent. The Glenco
3.2. Reagents
- 3.2.1. Carbon disulfide, reagent grade.
3.2.2. Isopropyl alcohol, reagent grade.
3.2.3. An internal standard, such as n-hexylbenzene, reagent grade.
3.2.4. Desorbing reagent - 1 uL internal standard/1 mL of 5% IPA in CS2.
3.2.5. Acetophenone, reagent grade.
3.2.6. Chromatographic quality nitrogen, hydrogen, and air.
3.3. Standard preparation
- 3.3.l. Standard of acetophenone is prepared by injecting 5 ul of
acetophenone into a
3.3.2. A calibration curve is prepared by making dilutions with the desorbing reagent.
3.4. Sample preparation
- 3.4.1 The front and back section of each sample are transferred
to separate
3.4.2. Each sample is desorbed with 1.0 mL of desorbing reagent.
3.4.3. The vials are sealed immediately and allowed to desorb for 30 minutes with occasional shaking.
3.5. Analysis
- 3.5.1. GC conditions
| Flow rates (mL/min) | Temperature (°C) | ||
| Nitrogen | - 25 | Injector | - 200 |
| Hydrogen | - 20 | Detector | - 250 |
| Air | - 300 | Column | - 150° |
| Injection size - 2.0 ul | |||
| Elution time - 1:4 minutes | |||
3.5.2. Chromatogram
See Figure 1.
3.5.3. Peak areas are measured by an integrator or other suitable means.
3.5.4. An internal standard procedure is used. The integrator is calibrated to report results in ppm for a 12 liter air sample after correction for desorption efficiency.
3.6. Interferences
- 3.6.l. Any compound having the general retention time of the
analyte of the internal standard used is an interference. Possible
interferences should be listed on the sample data sheet. GC
parameters should be adjusted if necessary so these interferences
will pose no problems.
3.6.2. Retention time data on a single column is not considered proof of chemical identity. Samples over the PEL should be confirmed by GC/Mass Spec or other suitable means.
3.7. Calculations
- 3.7.1. The equivalent ppm air concentration of analytical
standards based on 12 liter air sample, and 1 mL desorbing solution
is calculated in the following manner:
| X µg/mL (24.45) (1 mL)
(mw) (12 L) |
= ppm |
| where | X µg/mL | = | concentration of the analytical standard. |
| 24.45 | = | Molar volume (liter/mole) at 25°C and 760 mm Hg | |
| MW | = | Molecular weight = 120.15 | |
| 1 mL | = | Desorption volume | |
| 12 L | = | 12 liter air sample |
3.7.2. Since the integrator was calibrated to report results in ppm based on 12 L air sample, the following calculation is used:
| ppm analyte = | A - C
B / 12 |
| where | A | = | ppm on report |
| C | = | ppm on report for blank | |
| B | = | air volume of sample (liters) |
3.7.3. This calculation is done for each section of the sampling tube and the results added together.
3.8. Safety precautions
- 3.8.l. All handling of solvents should be done in a hood.
3.8.2. Avoid skin contact with all solvents.
3.8.3. Wear safety glasses at all times.
4. Recommendations for further study
- 4.1. Further work should be done on breakthrough.
4.2. Longer storage tests should be performed.
Figure 1
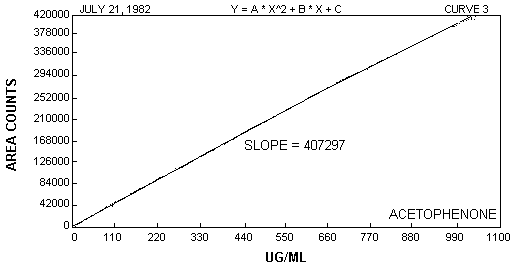
Figure 2
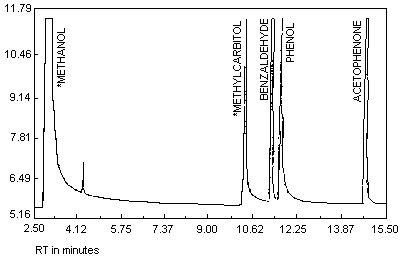
A bulk sample was analyzed by gas chromatography on a Varian 3400 with
a flame ionization detector. The analysis was for % methyl carbitol,
benzaldehyde, phenol, and acetophenone, with methanol as the solvent. The
column used was a 60 M
5. References
- 5.1. "The Condensed Chemical Dictionary", 10th ed.; Hawley, G.G.,
ed.; Van Norstrand Reinhold Company, New York, 1981; p. 9.
5.2. "The Merck Index", 9th ed.; Windholz, Martha, ed.; Merck and Company, Inc., New Jersey, 1976; p. 9.
5.3. "Spice Mill"; Katz, A.E.; 69, 1946; p. 46.
5.4. "Journal of Industrial Hygiene Toxicology"; Smyth, H.F. Jr., and Carpenter, C.P.; 30, 1948; p. 63.